The psychedelic renaissance represents a $3.88 billion market opportunity that extends far beyond biotech into energy-intensive infrastructure and commodity supply chains. The shift from $10,000-per-gram chemical synthesis to sub-$100-per-gram biosynthesis isn't just about cost—it's about energy efficiency, supply chain resilience, and the buildout of an entirely new pharmaceutical infrastructure. Every milligram of pharmaceutical-grade psilocybin represents a complex supply chain of rare earth catalysts, L-tryptophan precursors, energy-intensive GMP facilities, and cold chain logistics. The investment thesis: while Wall Street chases biotech headlines, sophisticated investors should focus on the picks-and-shovels infrastructure providers that will enable the entire industry.
The Premise: A Wellness Revolution with an Industrial Footprint
Psychedelic microdosing has evolved from counter-culture curiosity into a serious contender for cognitive enhancement in professional settings. Sub-perceptual doses of compounds like psilocybin—typically one-tenth to one-twentieth of a recreational dose—are showing promise for enhancing creativity, focus, and emotional resilience without hallucinogenic effects. Early research reports improvements in problem-solving ability by up to 20%, and the cognitive enhancement market is projected to reach $1.5 billion by 2027.
But here's what the wellness headlines miss: this isn't just a pharmaceutical development story. It's an industrial buildout on the scale of the early biotech revolution, requiring massive investments in energy-intensive infrastructure, specialized commodity supply chains, and logistics networks that don't yet exist at commercial scale. The companies that solve the energy, commodity, and infrastructure challenges will capture value that dwarfs the biotech players themselves.
The Energy Economics of Consciousness: Three Production Pathways
The path from laboratory curiosity to mainstream cognitive enhancer requires solving a fundamental industrial challenge: how do you produce pharmaceutical-grade psilocybin at scale, at cost, and with acceptable energy efficiency? Three competing production methods are emerging, each with radically different energy footprints and commodity requirements.
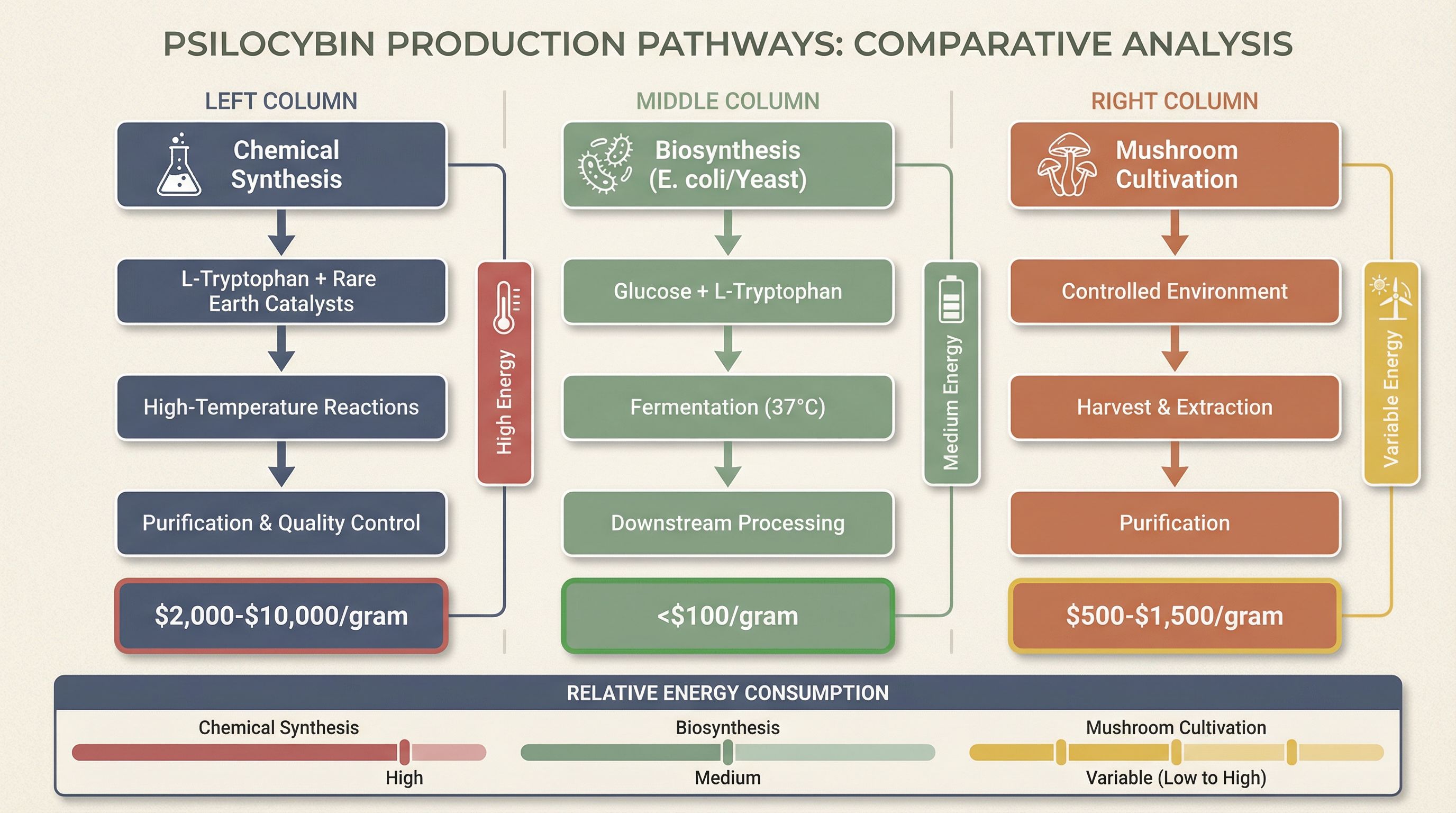
Chemical Synthesis: The Energy-Intensive Incumbent
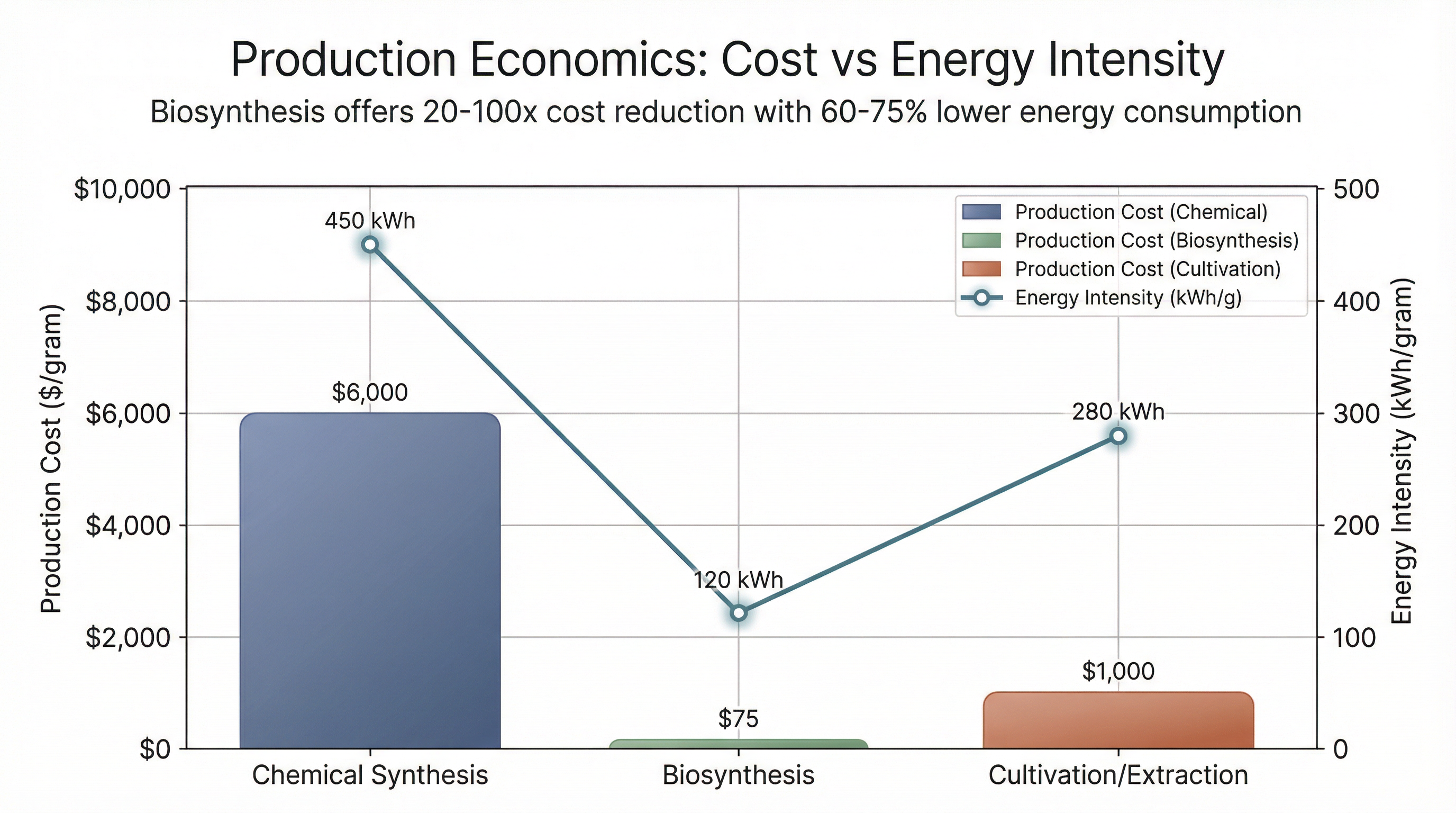
Traditional chemical synthesis of psilocybin carries a staggering price tag: $2,000 to $10,000 per gram as of 2025. This isn't just about chemical complexity—it's about energy. Chemical synthesis requires high-temperature reactions, specialized equipment (reactors, purification systems), extensive solvent recovery, and waste treatment. The process demands rare earth and lanthanide-based catalysts for precision drug synthesis, creating dependency on overseas suppliers (primarily China and India) for key chemical reagents.
A 2026 breakthrough offers a glimpse of the future: researchers developed a blue-light-powered iron catalyst that replaces rare metals in drug synthesis, potentially reducing both cost and energy consumption. But even with these advances, chemical synthesis remains the most energy-intensive production pathway, requiring continuous operation of climate-controlled GMP (Good Manufacturing Practices) facilities with stringent air filtration, temperature control, and quality assurance protocols.
Biosynthesis: The Energy-Efficient Disruptor
The game-changer is biosynthesis: using genetically engineered E. coli or yeast to produce psilocybin from glucose. Research teams have demonstrated "homebrew style" production in recombinant E. coli strains, producing biologically relevant quantities without the energy intensity of chemical synthesis. This method requires fermentation infrastructure—temperature-controlled tanks, agitation systems, downstream processing—but the overall energy footprint is dramatically lower than chemical synthesis.
The key commodity input is L-tryptophan, the precursor amino acid with an estimated annual market potential of 11,000 tons. Biosynthesis routes consume electricity for fermentation equipment and climate control, but eliminate the need for rare earth catalysts and high-temperature reactions. The economic advantage is stark: production costs can drop below $100 per gram, a 20x to 100x reduction compared to chemical synthesis.
Cultivation and Extraction: The Organic Alternative
The third pathway—cultivating Psilocybe mushrooms and extracting psilocybin—carries its own energy profile. Cultivation requires controlled-environment agriculture: HVAC systems for temperature and humidity control, lighting (if not natural), and water management. Extraction methods (ultrasonic, solvent-based) add additional energy consumption, though typically less than chemical synthesis.
Interestingly, clinical studies suggest mushroom extract may be more effective than synthetic psilocybin, and patient reports describe organic forms as "more alive and vibrant" compared to synthetic versions. This efficacy premium could justify the higher energy costs of cultivation, particularly if renewable energy sources (solar, wind) power the growing facilities. The energy transition angle here is compelling: solar-powered mushroom cultivation facilities could produce pharmaceutical-grade psilocybin with near-zero carbon footprint.
The Infrastructure Imperative: GMP Facilities, Cold Chains, and Compliance
Regardless of production method, bringing psychedelic cognitive enhancers to market requires navigating a gauntlet of infrastructure challenges that are fundamentally energy and commodity stories.
GMP Pharmaceutical Facilities: The Energy Hogs
Good Manufacturing Practices (GMP) are mandatory for clinical trials and commercial production of psychedelic drugs. FDA and DEA compliance demands clean room standards with extraordinary energy consumption: continuous air filtration, precise temperature and humidity control, and extensive quality control testing. A single GMP facility can consume megawatts of power annually, with HVAC systems representing the largest energy draw.
The current bottleneck: there are insufficient DEA-licensed psychedelic production facilities to meet projected demand. Building new GMP facilities requires capital expenditures in the tens of millions of dollars, with ongoing energy costs that rival semiconductor fabrication plants. Companies that can finance and operate these facilities efficiently—particularly those integrating renewable energy—will command pricing power as the market scales.
Pharmaceutical-grade psilocybin requires temperature-controlled storage and refrigerated transport from production to end-user. This cold chain infrastructure demands continuous refrigeration, representing a persistent energy cost that scales with market adoption. Tamper-proof, FDA/DEA-compliant packaging systems add additional complexity and cost.
The distribution challenge is particularly acute for a Schedule I controlled substance: secure, compliant cold chain logistics networks don't yet exist at the scale required for a multi-billion-dollar market. Companies that build this infrastructure—refrigerated warehouses, secure transport fleets, temperature-monitoring systems—will capture value as essential middlemen in the psychedelic supply chain.
The L-Tryptophan Bottleneck
Every production pathway—chemical synthesis, biosynthesis, and even mushroom cultivation—depends on L-tryptophan, either as a direct chemical precursor or as a metabolic pathway input. With an estimated 11,000-ton annual market potential, L-tryptophan supply chains are poised for massive expansion. Current production relies heavily on fermentation by specialized microorganisms, creating a nested dependency: the psychedelic industry depends on L-tryptophan producers, who themselves depend on glucose feedstocks and fermentation infrastructure.
Investors should watch L-tryptophan producers and fermentation equipment manufacturers as critical chokepoints in the psychedelic supply chain. Disruptions in L-tryptophan supply—whether from feedstock shortages, energy price spikes, or geopolitical tensions affecting overseas suppliers—could ripple through the entire psychedelic industry.
Market Implications: The $3.88 Billion Infrastructure Play
The psychedelic drug market reached $3.88 billion in 2025, with cognitive enhancement representing a $1.5 billion subset by 2027. But these headline numbers obscure the deeper infrastructure opportunity. The real value creation isn't in the biotech companies developing novel compounds—it's in the picks-and-shovels providers enabling the entire industry.
The Professional Productivity Premium
The target market for cognitive enhancement is vast: high-stress, high-demand professionals in technology, finance, creative industries, and medicine. These professionals represent trillions of dollars in economic output, and even modest improvements in cognitive performance translate to immense value. If microdosing can reliably enhance focus by 20%, reduce stress, and boost creativity, the return on investment for individuals and organizations could redefine corporate performance metrics.
This demand creates a pull effect throughout the supply chain. As professional adoption scales, the infrastructure requirements multiply: more GMP facilities, more fermentation capacity, more L-tryptophan production, more cold chain logistics. The companies positioned at these infrastructure chokepoints will capture disproportionate value as the market expands.
The Convergence of Biotech, Wellness Tech, and Industrial Infrastructure
The psychedelic renaissance sits at the intersection of three massive industries: biotech (drug development), wellness tech (digital therapeutics, biometric tracking), and industrial infrastructure (chemical production, logistics). This convergence creates opportunities for sophisticated investors to build portfolios that capture value across the entire value chain.
Biotech companies will develop novel compounds and delivery mechanisms. Wellness tech platforms will integrate biometric tracking, personalized dosing algorithms, and digital therapeutics to complement microdosing regimens. But the unglamorous infrastructure providers—fermentation equipment manufacturers, pharmaceutical-grade HVAC systems, L-tryptophan producers, cold chain logistics companies—will enable the entire ecosystem.
This level of multi-dimensional analysis—connecting biotech innovation to energy infrastructure and commodity supply chains—represents the discretionary research approach that uncovers second-order investment opportunities. For investors who also want systematic, emotion-free exposure to broad market trends, Vetta's V-Rank Alpha provides a complementary strategy: two proprietary algorithms select 20-40 positions from S&P 500/400 constituents monthly, delivering 1,845% returns since 2005. While we focus on uncovering hidden structural dynamics in emerging industries, Vetta's algorithmic approach captures technology shifts and sector rotations systematically, removing emotional bias from security selection.
The Investment Thesis: Picks, Shovels, and Infrastructure
The psychedelic gold rush creates distinct investment opportunities across three categories: biotech innovators, infrastructure enablers, and commodity suppliers. The highest-conviction plays combine multiple advantages: energy efficiency, supply chain control, and regulatory positioning.
Biotech's Vanguard: The Pharmaceutical Play
Compass Pathways (CMPS) represents the focused biotech bet. The company is advancing psilocybin therapy through clinical trials with a clear regulatory pathway. Their COMP360 psilocybin formulation targets treatment-resistant depression, a massive unmet medical need. The investment case hinges on clinical trial success and FDA approval, but the company benefits from first-mover advantage in establishing GMP production capabilities and navigating DEA licensing.
The risk: biotech is inherently binary. Clinical trial failures, regulatory setbacks, or competitive pressures from biosynthesis-enabled competitors could crater valuations. Compass's production method (likely chemical synthesis given their early-stage development) exposes them to higher energy costs and commodity price volatility compared to future biosynthesis competitors.
World’s First Safe AI-Native Browser
AI should work for you, not the other way around. Yet most AI tools still make you do the work first—explaining context, rewriting prompts, and starting over again and again.
Norton Neo is different. It is the world’s first safe AI-native browser, built to understand what you’re doing as you browse, search, and work—so you don’t lose value to endless prompting. You can prompt Neo when you want, but you don’t have to over-explain—Neo already has the context.
Why Neo is different
Context-aware AI that reduces prompting
Privacy and security built into the browser
Configurable memory — you control what’s remembered
As AI gets more powerful, Neo is built to make it useful, trustworthy, and friction-light.
Infrastructure Enablers: The Unglamorous Winners
The highest-conviction infrastructure plays are harder to isolate in public markets, as many are divisions within larger industrial conglomerates. But the categories are clear:
Fermentation Equipment Manufacturers: Companies producing industrial-scale bioreactors, fermentation tanks, and downstream processing equipment will see surging demand as biosynthesis becomes the dominant production method. Look for exposure through pharmaceutical equipment manufacturers with fermentation capabilities.
Pharmaceutical-Grade HVAC Systems: Specialized climate control systems for GMP facilities represent a recurring revenue stream as new psychedelic production facilities come online. Companies with expertise in clean room HVAC for pharmaceutical and semiconductor applications are positioned to capture this demand.
L-Tryptophan Producers: Direct exposure to the critical precursor commodity. As psychedelic production scales, L-tryptophan demand will surge beyond current market capacity. Producers with fermentation expertise and the ability to rapidly expand capacity will command pricing power.
Cold Chain Logistics: Refrigerated warehousing and transport providers with DEA compliance capabilities will become essential middlemen. The security and regulatory requirements create high barriers to entry, protecting margins for established players.
The Continuous Flow Revolution
A transformative technology is emerging that could reshape the entire production landscape: continuous flow manufacturing. This advanced manufacturing technique reduces production time from months to days, dramatically lowering energy consumption and capital requirements. Companies that successfully implement continuous flow for psychedelic synthesis will achieve cost structures that make chemical synthesis competitive with biosynthesis, while maintaining the purity advantages of synthetic compounds.
The energy transition angle is compelling: continuous flow manufacturing is inherently more energy-efficient than batch processing, and can be more easily integrated with intermittent renewable energy sources (solar, wind). Pharmaceutical companies investing in continuous flow capabilities are positioning for a future where energy costs and carbon footprints become competitive differentiators.
Investment Strategies
The following examples are for educational purposes only and do not constitute investment advice. Consult with a qualified financial advisor before making any investment decisions.
Bull Put Spread: Compass Pathways (CMPS)
Thesis: Compass Pathways' focused approach to psilocybin therapy, combined with first-mover advantage in GMP production and regulatory navigation, positions the company to capture significant market share as psychedelic treatments gain FDA approval. Clinical trial progress and regulatory momentum support a bullish outlook.
Example Structure (Educational):
•Sell 1 CMPS $8 Put (expires in 90 days)
•Buy 1 CMPS $6 Put (expires in 90 days)
•Max Profit: Premium collected (if CMPS stays above $8)
•Max Loss: $2 per share minus premium (if CMPS falls below $6)
•Breakeven: $8 minus net premium received
Rationale: This structure profits if CMPS stays above $8, reflecting confidence in the company's clinical progress and regulatory pathway. The defined risk ($2 spread) limits downside while generating income from the company's first-mover positioning. The strategy acknowledges biotech volatility while expressing a bullish view on the psychedelic therapy opportunity.
Bear Call Spread: PureTech Health (PRTCY)
Thesis: PureTech Health's diversified approach dilutes exposure to the psychedelic opportunity, spreading capital across multiple therapeutic areas without the focused execution required to win in the rapidly evolving psychedelic market. While diversification reduces risk, it also limits upside in a sector where first-mover advantage and production expertise are critical.
Example Structure (Educational):
•Sell 1 PRTCY $30 Call (expires in 90 days)
•Buy 1 PRTCY $35 Call (expires in 90 days)
•Max Profit: Premium collected (if PRTCY stays below $30)
•Max Loss: $5 per share minus premium (if PRTCY rises above $35)
•Breakeven: $30 plus net premium received
Rationale: This structure profits if PRTCY stays below $30, reflecting the thesis that diversified biotech platforms will underperform focused psychedelic players as the market matures. The defined risk ($5 spread) protects against unexpected positive catalysts while capitalizing on the company's lack of competitive focus in psychedelic production and infrastructure.
Why This Pairing Works
The strategies isolate the "focus vs. diversification" factor in psychedelic biotech. Compass (focused) should outperform as the market rewards specialized expertise in production, regulatory navigation, and clinical development. PureTech (diversified) should underperform as capital allocation across multiple therapeutic areas dilutes returns from the psychedelic opportunity. This creates a sector-neutral position focused specifically on business model efficiency rather than directional biotech bets.
The psychedelic infrastructure buildout faces significant headwinds that could delay or derail the investment thesis.
Regulatory Labyrinth
Psychedelics remain Schedule I controlled substances in most jurisdictions, creating extraordinary regulatory hurdles for production, distribution, and clinical use. FDA approval pathways exist, but timelines are uncertain and subject to political pressures. DEA licensing for production facilities is a slow, complex process that limits supply even as demand grows.
Public perception remains mixed, with stigma from decades of drug war rhetoric competing against emerging clinical evidence. Regulatory setbacks—whether from adverse events in clinical trials, political backlash, or enforcement actions—could freeze the market overnight.
Safety and Dosing Precision
Microdosing requires extraordinary precision: the difference between a sub-perceptual dose and a hallucinogenic dose is narrow, and individual sensitivity varies widely. Standardization of dosing, delivery mechanisms, and quality control will be critical for mainstream adoption. Any high-profile adverse events—particularly in professional settings—could trigger regulatory crackdowns and public backlash.
Long-term effects of repeated microdosing remain unknown. While early research is promising, the possibility of unforeseen health consequences creates tail risk for the entire industry.
Supply Chain Vulnerabilities
The psychedelic supply chain's dependency on overseas suppliers for key chemical reagents (particularly from China and India) creates geopolitical risk. Trade tensions, export restrictions, or supply disruptions could spike costs and delay production. L-tryptophan supply constraints could bottleneck the entire industry if demand surges faster than production capacity can scale.
Energy price volatility directly impacts production economics, particularly for energy-intensive chemical synthesis and GMP facility operations. Companies without energy hedging strategies or renewable energy integration will face margin pressure as energy costs fluctuate.
Future Outlook: The 2-5 Year Horizon
The next 2-5 years will determine whether the psychedelic cognitive enhancement revolution achieves mainstream adoption or remains a niche market. Several key inflection points will shape the trajectory.
The Tipping Point: Mainstream Acceptance
FDA approval of psilocybin therapy for treatment-resistant depression would catalyze the entire market, legitimizing psychedelic compounds and accelerating research into cognitive enhancement applications. Corporate adoption of microdosing protocols—whether formally sanctioned or tacitly accepted—would create massive demand from the professional productivity market.
The wellness tech integration will be critical: platforms that seamlessly combine biometric tracking, personalized dosing algorithms, and digital therapeutics will drive adoption by making microdosing accessible, safe, and measurable. AI-powered optimization of dosing schedules based on individual biology could unlock the full cognitive enhancement potential.
The Personalized Brain: Precision Neuro-Optimization
The future of cognitive enhancement isn't one-size-fits-all microdosing—it's precision neuro-optimization tailored to individual biology, work patterns, and cognitive goals. Wearable devices tracking sleep, stress, focus, and creativity will feed AI algorithms that optimize dosing schedules in real-time. This convergence of psychedelics, wellness tech, and AI represents a multi-hundred-billion-dollar opportunity that extends far beyond the psychedelic compounds themselves.
The infrastructure required for this personalized future—cloud computing for AI algorithms, wearable device manufacturing, secure health data platforms—represents another layer of investment opportunities adjacent to the core psychedelic market.
The Final Word: Follow the Infrastructure
The psychedelic renaissance is real, but the investment opportunity isn't where most people are looking. While biotech stocks capture headlines, the real value creation is happening in the unglamorous infrastructure that enables the entire industry: fermentation equipment, GMP facilities, L-tryptophan supply chains, cold chain logistics, and continuous flow manufacturing.
As an investor, your job is to look past the wellness narratives and dig into the industrial reality. Find the companies that talk about their fermentation capacity, their energy efficiency, their supply chain resilience. These are the companies that understand the true nature of the opportunity: not just a pharmaceutical development story, but an industrial buildout on the scale of the early biotech revolution.
The psychedelic gold rush is here. The winners will be those who bring the picks and shovels…
Until Next Time, Good Luck and Good Investing!

C.D. Lawrence, Senior Analyst, Solar Kitties Research
Source Key
[1] ScienceDirect (2025): [Is there mush-room to improve the environmental sustainability of psilocybin production?](https://www.sciencedirect.com/science/article/pii/S2212982025001210 ) - Reports estimated cost of $2,000-$10,000 USD per gram for synthetically produced psilocybin.
[2] NIH/PMC (2021): [Homebrewed psilocybin: can new routes for pharmaceutical production improve supply?](https://pmc.ncbi.nlm.nih.gov/articles/PMC8806991/ ) - Demonstrates psilocybin production in recombinant E. coli in homebrew style environment.
[3] Wiley Online Library (2023): [Biosynthesis of psilocybin and its nonnatural derivatives](https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/10.1002/bit.28480 ) - Reports bacterial system for de novo biosynthesis of psilocybin from glucose.
[4] MDPI Pharmaceuticals (2025): [Extraction Yields of Psilocybin and Psilocin](https://www.mdpi.com/1424-8247/18/3/380 ) - Systematic review of extraction and quantification methods.
[5] Nature (2024): [Effect of chemically synthesized psilocybin and psychedelic mushroom extract](https://www.nature.com/articles/s41380-024-02477-w ) - Shows mushroom extract more effective than chemical psilocybin.
[6] SciTechDaily (2026): [Blue-Light Iron Breakthrough Could Make Drug Production Cheaper](https://scitechdaily.com/this-blue-light-iron-breakthrough-could-make-drug-production-cheaper/ ) - Reports blue-light-powered iron catalyst replacing rare metals in drug synthesis.
[7] Wiley Online Library (2021): [Rare-Earth Incorporated Alloy Catalysts](https://advanced.onlinelibrary.wiley.com/doi/10.1002/adma.202005988 ) - Reviews rare-earth alloys as catalytic materials in energy conversion.
[8] ScienceDirect (2011): [Enzymatic synthesis of l-tryptophan](https://www.sciencedirect.com/science/article/abs/pii/S0960852410015312 ) - Estimates 11,000 ton annual market potential for L-tryptophan.
[9] Packaging Digest (2025): [Psychedelic Drug Market Spurs Packaging Challenges](https://www.packagingdigest.com/pharmaceutical-packaging/-6-33-psychedelic-drug-market-spurs-packaging-challenges ) - Reports $3.88 billion psychedelic drug market.
[10] Psychedelic Alpha: [The Psychedelic Value Chain](https://psychedelicalpha.com/news/the-psychedelic-value-chain ) - Analysis of manufacturing and supply chain for psychedelic drugs.
[11] C&EN (2025): [Raw materials from overseas fuel the production of medicines](https://cen.acs.org/pharmaceuticals/Raw-materials-overseas-fuel-production/103/web/2025/10 ) - Reports key chemicals often come from China or India.
[12] APIIC (2025): [Building a Resilient Domestic Drug Supply Chain](https://apicenter.org/wp-content/uploads/2025/03/APIIC-White-Paper-2025-Building-a-Resilient-Domestic-Drug-Supply-Chain.pdf ) - Discusses continuous flow manufacturing reducing production time from months to days.
Additional Context Sources:
Disclaimer
This article is for educational and informational purposes only and does not constitute financial, investment, or trading advice. The investment strategies discussed are hypothetical examples intended to illustrate concepts related to psychedelic drug production infrastructure and supply chains. Options trading involves substantial risk and is not suitable for all investors. Past performance is not indicative of future results. Readers should conduct their own research and consult with a qualified financial advisor before making any investment decisions. The author and Solar Kitties Research are not registered investment advisors and do not provide personalized investment recommendations.
About Solar Kitties Research
Solar Kitties is an independent research platform exploring the intersection of energy transition, commodity markets, and investment strategy. We focus on uncovering hidden structural dynamics that drive long-term value creation in emerging industries and related sectors.
For technology disruption analysis and algorithmic investing insights, subscribe to our sister publication "The Long & Short of It" from Vetta Investments.
Follow us for more deep-dive analysis at solarkitties.com
Subscribe to Chip in - Pay What You Can to read the rest.
Become a paying subscriber of Chip in - Pay What You Can to get access to this post and other subscriber-only content.
Upgrade



